Acute Intermittent Porphyria Market Introduction
The Acute Intermittent Porphyria (AIP) market represents a critical area within the rare disease and orphan drug segments of the healthcare industry. AIP is a rare metabolic disorder that affects the production of heme, the oxygen-binding prosthetic group of hemoglobin, due to a deficiency of the enzyme porphobilinogen deaminase. The disease is characterized by potentially life-threatening acute attacks with symptoms including severe abdominal pain, neurological complications, and mental disturbances. Due to its low prevalence and high disease burden, AIP is categorized under orphan indications, which makes its treatment market niche but medically significant.
Recent advancements in genetic research, diagnostic capabilities, and therapeutic development have increased global awareness about AIP. These developments have encouraged pharmaceutical research and investment into novel treatments and management solutions, with a focus on precision therapies, early diagnosis, and preventative strategies. Moreover, governments and regulatory bodies have extended incentives such as market exclusivity and grants for orphan drug development, which has fueled interest among biotech innovators and healthcare providers.
With increasing support from patient advocacy groups, combined with improved healthcare access in emerging economies, the AIP market is witnessing a gradual yet steady expansion. However, challenges remain in terms of limited therapeutic options, delayed diagnoses, and high treatment costs, which restrict broader market penetration. Furthermore, the episodic and unpredictable nature of AIP presents difficulties in consistent monitoring and management.
The market for Acute Intermittent Porphyria is thus poised at a crucial juncture where unmet medical needs intersect with technological innovation and regulatory encouragement. As stakeholders strive for improved outcomes, the market holds potential for breakthroughs that can transform patient care and redefine the standards of treatment for rare genetic disorders.
Acute Intermittent Porphyria Market Definition
Acute Intermittent Porphyria (AIP) is a rare, autosomal dominant genetic disorder that falls under the broader classification of hepatic porphyrias. It results from a partial deficiency in the enzyme porphobilinogen deaminase (PBGD), also known as hydroxymethylbilane synthase, which is involved in the heme biosynthesis pathway. This enzymatic insufficiency leads to the accumulation of neurotoxic intermediates such as porphobilinogen (PBG) and delta-aminolevulinic acid (ALA), particularly during metabolic stress, triggering acute neurovisceral attacks.
AIP typically presents in adolescence or early adulthood and is characterized by episodic symptoms rather than continuous manifestations. Common symptoms include intense abdominal pain, vomiting, constipation, hypertension, tachycardia, seizures, and psychiatric symptoms like anxiety or hallucinations. If untreated, severe attacks may result in paralysis or death. Although AIP is rare, with an estimated prevalence of 1–10 per 100,000 people, it is often underdiagnosed due to its non-specific and variable symptoms.
Diagnosis of AIP involves clinical assessment, biochemical testing for elevated urinary ALA and PBG levels, and genetic testing to confirm mutations in the HMBS gene. Because AIP is hereditary, family screening can be critical for identifying asymptomatic carriers. Management of the disease involves acute treatment during attacks and long-term preventive strategies to avoid triggers such as certain medications, infections, hormonal fluctuations, and fasting.
Therapeutic approaches include intravenous hemin for acute attacks, pain management, and lifestyle adjustments. In recent years, emerging RNA interference therapies and gene-targeted treatments are under investigation or development, aiming to provide more targeted, disease-modifying effects. Despite advancements, AIP remains a challenging condition, requiring interdisciplinary care approaches involving neurologists, geneticists, hepatologists, and psychiatrists.
As a rare but impactful disorder, AIP commands attention from the medical community, policy-makers, and pharmaceutical developers alike, forming a distinct yet vital segment of the rare disease therapeutics market.
Acute Intermittent Porphyria Market Scope & Overview
The Acute Intermittent Porphyria (AIP) market encompasses a specialized segment of the rare disease landscape, focusing on the diagnosis, management, and treatment of this genetic metabolic disorder. Given the episodic nature of AIP and its life-threatening complications, the scope of the market extends beyond acute care to include chronic disease management, genetic counseling, diagnostic services, and preventive interventions.
Market participants range from biotechnology firms developing targeted therapies to diagnostic laboratories offering genetic and biochemical testing. Healthcare providers, research institutions, and patient advocacy organizations also play integral roles in expanding access and improving awareness. As therapeutic innovations gain traction, particularly those targeting the genetic and enzymatic roots of AIP, the market is shifting toward precision medicine and long-term disease control.
Geographically, the market shows stronger activity in developed regions with advanced healthcare infrastructures, such as North America and Europe. However, rising healthcare investments and increasing diagnostic capabilities in emerging markets are contributing to global expansion. Technological advancements in next-generation sequencing and RNA-based therapies have enhanced the ability to diagnose and treat AIP more effectively, broadening the market's scope.
The patient base remains limited due to the rarity of the disease, but the high unmet need and critical nature of care position AIP as a high-value therapeutic area. Regulatory incentives, such as orphan drug designation, fast-track approvals, and extended market exclusivity, further broaden the scope for pharmaceutical innovation and investment.
Overall, the AIP market represents a convergence of rare disease management, cutting-edge biotechnology, and regulatory support. While small in volume, it holds significant value in terms of patient outcomes, therapeutic innovation, and long-term healthcare impact, forming a niche yet promising frontier in the global biopharmaceutical ecosystem.
Acute Intermittent Porphyria Market Size
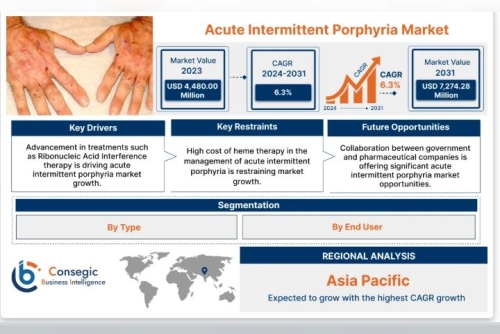
Acute intermittent porphyria market size is estimated to reach over USD 7,274.28 Million by 2031 from a value of USD 4,480.00 Million in 2023, growing at a CAGR of 6.3% from 2024 to 2031.
Acute Intermittent Porphyria Market Segmental Analysis
By Type
Treatment· Gonadotropin-releasing Hormone Analogues
· Ribonucleic Acid Interference therapy
· Others
Diagnosis· Urine Test
· Genetic Testing
· Others
By End-User
Hospitals Clinics Research Centers OthersAcute Intermittent Porphyria Market Key Industry Drivers & Trends
The Acute Intermittent Porphyria market is driven by a confluence of medical urgency, technological progress, and policy-level incentives. One of the most significant drivers is the high unmet need in effective and sustainable treatment options. AIP patients suffer from unpredictable, potentially life-threatening attacks, often requiring emergency interventions. This clinical burden has propelled demand for better therapeutic solutions.
Technological Advancements in genetic testing and molecular diagnostics are major market enablers. The adoption of next-generation sequencing (NGS) and RNA interference technologies has revolutionized both diagnosis and treatment development. These tools allow for earlier diagnosis, carrier detection, and gene-targeted therapies, significantly improving disease management strategies.
Regulatory Support through orphan drug incentives, fast-track designations, and rare disease policies are encouraging pharmaceutical investment. These policies reduce R&D risk and offer extended market exclusivity, fueling innovation in a space that traditionally faced high entry barriers due to the low patient population.
Increased Awareness and Advocacy from patient groups and rare disease organizations are leading to improved diagnosis rates and better patient outcomes. Educational campaigns and collaboration between stakeholders are fostering a more supportive ecosystem for AIP patients.
Trends in personalized medicine and biologics are shaping the treatment landscape. RNA-based therapies targeting ALAS1 enzyme expression are gaining attention for their ability to prevent recurrent attacks. Additionally, long-term prophylactic approaches are emerging as preferred treatment models over episodic management.
Challenges such as limited treatment access in low-income regions, high drug costs, and underdiagnosis continue to restrain market expansion. However, increased global focus on rare diseases is expected to mitigate these issues over time.
Collectively, these drivers and trends are reshaping the AIP market, transitioning it from reactive care to proactive, preventive, and personalized health solutions.
Acute Intermittent Porphyria Market Regional Analysis
The Acute Intermittent Porphyria (AIP) market demonstrates a geographically varied landscape, with market maturity and patient access differing significantly across regions.
North America holds the largest share of the AIP market, driven by robust healthcare infrastructure, high diagnostic capabilities, and strong presence of research institutions. Governmental support for orphan drug development and high awareness levels contribute to a favorable environment for therapeutic innovation. The U.S., in particular, leads in clinical trials and access to novel treatments.
Europe follows closely, benefiting from centralized healthcare systems, widespread adoption of genetic testing, and consistent policy support for rare diseases. Countries like Germany, the U.K., and France are at the forefront of diagnostics and patient support initiatives. The region’s collaborative rare disease networks enhance access and standardize care practices.
Asia Pacific is an emerging market with substantial growth potential. Although AIP awareness and diagnosis are relatively low compared to Western markets, improving healthcare infrastructure, growing investments in genomics, and increased medical tourism are contributing to its expansion. Countries such as Japan, China, and South Korea are investing in rare disease research and gradually integrating AIP care into broader healthcare services.
Latin America and the Middle East & Africa represent underpenetrated markets, primarily constrained by limited healthcare access and diagnostic capabilities. However, increasing collaborations with global health organizations and patient advocacy efforts are gradually opening up these regions to better diagnosis and care.
Regional disparities in prevalence reporting, treatment availability, and healthcare reimbursement systems highlight the need for a globalized approach to AIP care. Strategic alliances, government support, and education initiatives are crucial in addressing regional gaps. Overall, the global AIP market is witnessing a slow but consistent expansion, with regional markets at various stages of development and opportunity.
Acute Intermittent Porphyria Market Key Players
Alnylam Pharmaceuticals, Inc. (United States)
Recordati Rare Diseases (Italy)
Sanofi (France)
Clinuvel Pharmaceuticals Ltd (Australia)
Apellis Pharmaceuticals (United States)
Quest Diagnostics (United States)
Mayo Clinic Laboratories (United States)
Bachem (Switzerland)
Takeda Pharmaceutical Company Limited (Japan)
Ipsen Pharma (France)
Contact Us:
Consegic Business intelligence
Email : [email protected]
Sales : [email protected]