Capecitabine Market Report
Introduction
The Capecitabine market
is witnessing significant growth, driven by its pivotal role in oncology,
particularly in the treatment of colorectal, breast, and gastric cancers. As a
widely adopted oral chemotherapy agent, Capecitabine offers a more convenient
alternative to traditional intravenous drugs, making it a preferred choice
among patients and healthcare providers. Its ability to selectively target
tumor cells while minimizing systemic toxicity has contributed to its
widespread acceptance across global healthcare systems. With cancer incidence
on the rise worldwide, the demand for effective, accessible, and less invasive
treatment options like Capecitabine continues to climb.
The market is influenced by advancements in drug delivery technologies, growing awareness regarding cancer treatment options, and increasing healthcare expenditure, particularly in developing regions. Moreover, the aging global population and the rising prevalence of cancer are creating a robust demand for Capecitabine. Additionally, ongoing research efforts and clinical trials aimed at expanding its indications and enhancing its efficacy are expected to contribute to market growth.
Despite its therapeutic advantages, the Capecitabine market faces challenges such as adverse side effects, high treatment costs, and regulatory hurdles. However, the push for generic alternatives and biosimilar development is mitigating some of these concerns, providing cost-effective options to a broader patient base.
The market's dynamics are also shaped by strategic collaborations among pharmaceutical companies, research institutions, and healthcare organizations aiming to streamline drug development and improve treatment outcomes. As healthcare infrastructure continues to improve globally, particularly in emerging economies, the Capecitabine market is projected to grow steadily. In summary, the Capecitabine market is poised for continued expansion, fueled by increasing cancer burden, patient-centric treatment approaches, and supportive regulatory frameworks that promote access to essential oncology drugs.
Definition
Capecitabine is an
orally-administered chemotherapeutic agent classified as an antimetabolite. It
is a prodrug of 5-fluorouracil (5-FU), meaning it is metabolized within the
body to become the active cytotoxic compound. Once administered, Capecitabine
undergoes enzymatic conversion to 5-FU specifically in tumor tissues, where it
inhibits DNA synthesis and impairs cancer cell proliferation. This selective
activation reduces systemic toxicity and enhances patient tolerance compared to
traditional chemotherapies.
Designed for the treatment of various solid tumors, Capecitabine is commonly used in colorectal, breast, and gastric cancer therapies. Its oral administration offers a convenient alternative to intravenous 5-FU, reducing hospital visits and improving patient quality of life. Capecitabine is often used as a monotherapy or in combination with other chemotherapeutic agents and targeted therapies, depending on the cancer type, stage, and treatment regimen.
Pharmacologically, Capecitabine disrupts the synthesis of thymidylate, an essential component of DNA, thus interfering with the cell cycle and inducing apoptosis in rapidly dividing tumor cells. The drug's effectiveness, coupled with its targeted mechanism of action, has made it a mainstay in oncology protocols.
Capecitabine’s development marked a shift toward more patient-friendly chemotherapy options, offering improved convenience without compromising efficacy. Its approval across multiple countries for specific cancer indications reflects the global consensus on its therapeutic value. However, it can cause side effects such as hand-foot syndrome, gastrointestinal disturbances, and hematologic toxicity, which require careful patient monitoring and dose adjustments.
The definition of Capecitabine in the market context extends beyond its pharmacological properties to encompass its role in modern cancer treatment strategies. As part of evolving oncologic therapies, Capecitabine continues to be integral in protocols aimed at enhancing survival rates, minimizing side effects, and providing outpatient-friendly cancer care.
Scope & Overview
The Capecitabine market spans a broad landscape that intersects the fields of
oncology, pharmaceutical innovation, and patient-centered treatment solutions.
The drug’s scope is largely defined by its therapeutic applications, which
include treating colorectal, breast, and gastric cancers—three of the most
prevalent forms of cancer globally. Capecitabine’s formulation allows for oral
administration, positioning it as a convenient alternative to traditional intravenous
chemotherapy regimens. This convenience factor expands its market reach,
especially in outpatient and home care settings.
From a market perspective, Capecitabine has gained significant traction in both developed and emerging economies, where cancer care infrastructure is growing. The global increase in cancer incidence, coupled with rising healthcare expenditure, has amplified demand for effective and tolerable chemotherapy drugs like Capecitabine. The drug is widely integrated into treatment guidelines and protocols across various countries, enhancing its market penetration.
The overview of the Capecitabine market reflects a competitive landscape characterized by the presence of both branded and generic formulations. This competition has led to a gradual reduction in treatment costs, improving accessibility for a broader population. Moreover, continual clinical research and trials are expanding Capecitabine's potential use in other cancer types and in combination with immunotherapies and targeted agents, thereby enlarging its application spectrum.
Healthcare policy reforms that support generic drug approvals and reimbursements are playing a vital role in shaping the Capecitabine market. Additionally, digital healthcare advancements, such as telemedicine and remote patient monitoring, are supporting the outpatient use of oral chemotherapeutics.
In conclusion, the Capecitabine market’s scope is multidimensional, involving clinical efficacy, patient adherence, regulatory policies, and healthcare system evolution. It encompasses a dynamic and growing segment of the oncology pharmaceutical industry, with significant opportunities for innovation, expansion, and improved cancer care delivery worldwide.
Size
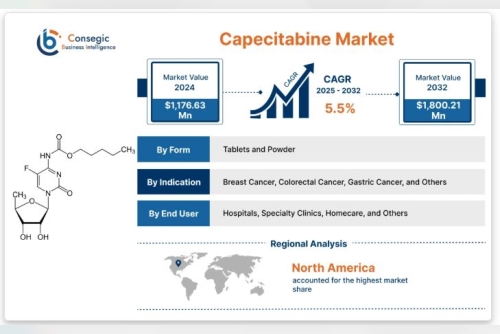
Consegic Business Intelligence analyzes that the capecitabine market size is growing with a healthy CAGR of 5.5% during the forecast period (2025-2032), and the market is projected to be valued at USD 1,800.21 Million by 2032 from USD 1,176.63 Million in 2024.
Segmental Analysis
By Form
Tablets and Powder
By Indication
Breast Cancer, Colorectal Cancer, Gastric Cancer, and Others
By End User
Hospitals, Specialty Clinics, Homecare, and Others
Key Industry Drivers &
Trends
The Capecitabine market is underpinned by several key drivers and evolving
trends that collectively influence its growth trajectory and competitive
landscape.
Major Market Drivers:
Rising Cancer Incidence: Increasing prevalence of cancers, particularly colorectal, breast, and gastric cancers, is a primary growth driver. With more patients being diagnosed annually, the demand for effective chemotherapeutic agents like Capecitabine is growing. Preference for Oral Chemotherapy: Capecitabine’s oral administration is significantly influencing its adoption. It offers convenience, reduces hospitalization costs, and aligns with patient-centered care trends, thereby driving market uptake. Healthcare Advancements and Accessibility: The expansion of oncology treatment centers, increased cancer screening, and government health initiatives—especially in developing nations—are enhancing access to medications like Capecitabine. Generic Drug Availability: Patent expirations have facilitated the production and approval of generic versions of Capecitabine, improving affordability and accessibility, which in turn propels market growth.Emerging Market Trends:
Combination Therapies: There is a rising trend of combining Capecitabine with targeted therapies and immunotherapies to enhance therapeutic efficacy, especially in resistant cancer types. Personalized Medicine: Increasing emphasis on tailoring treatments to individual patient profiles is driving demand for pharmacogenetic testing, influencing dosage and treatment planning for Capecitabine. Outpatient and Home-Based Treatment Models: With growing interest in remote care and telehealth, Capecitabine’s role in home-based cancer therapy is gaining momentum. Regulatory Streamlining: Faster drug approvals and efforts to support biosimilar development are encouraging innovation and rapid market entry.Together, these drivers and trends are shaping a forward-moving and patient-centric Capecitabine market. Stakeholders are leveraging technological, clinical, and policy-based developments to support market expansion, while addressing challenges such as side-effect management and resistance.
Regional Analysis
The Capecitabine market displays varied dynamics across major global regions,
reflecting differences in healthcare infrastructure, cancer prevalence,
regulatory frameworks, and treatment accessibility.
North America:
North America holds a significant share of the Capecitabine market, driven by
advanced healthcare systems, high cancer diagnosis rates, and strong
reimbursement policies. The United States, in particular, benefits from
widespread awareness, access to oncology specialists, and well-established clinical
protocols that include Capecitabine. Research and development efforts and
clinical trials further strengthen the region’s market presence.
Europe:
Europe follows closely, with countries such as Germany, the UK, France, and
Italy playing key roles. The region benefits from robust public healthcare
systems and standardized cancer treatment guidelines. Initiatives promoting
early diagnosis and access to generic drugs support the market. The focus on
cost-effective treatment options has increased the adoption of Capecitabine
across both Western and Eastern Europe.
Asia-Pacific:
Asia-Pacific is emerging as a highly lucrative market due to the rising cancer
burden, growing healthcare infrastructure, and increasing awareness of oral
chemotherapeutics. Countries like China, India, and Japan are witnessing
significant demand, propelled by population growth and improved access to
healthcare services. Government efforts to expand oncology care and the
presence of domestic pharmaceutical manufacturers are further supporting
regional growth.
Latin America and Middle
East & Africa:
These regions present promising growth opportunities, albeit from a smaller
base. In Latin America, countries such as Brazil and Mexico are investing in
cancer care infrastructure, improving access to essential drugs. In the Middle
East & Africa, market growth is supported by urbanization, international
aid programs, and the gradual expansion of healthcare services.
In summary, while mature markets lead in terms of value, emerging economies offer immense untapped potential. Regional variations in regulation, pricing, and access will continue to influence the overall performance and strategic direction of the Capecitabine market.
Key Players
Teva Pharmaceuticals, Inc, F. Hoffmann-La Roche AG, Mylan N.V., Hikma
Pharmaceuticals PLC, Cipla Inc., Dr. Reddy's Laboratories Ltd., Fresenius SE
& Co. KGaA, Sun Pharmaceutical Industries Limited, Genentech, Inc., and
Accord Healthcare Ltd.
Contact Us:
Consegic Business intelligence
Email : [email protected]
Sales : [email protected]