Dravet Syndrome Market Report
Introduction:
The Dravet Syndrome market
has witnessed significant advancements driven by evolving therapeutic needs and
increasing awareness surrounding rare and genetic epileptic disorders. Dravet
Syndrome, also known as severe myoclonic epilepsy of infancy (SMEI), is a
catastrophic form of epilepsy that begins in the first year of life and
presents with frequent and prolonged seizures. Due to the rarity and severity
of the condition, the market has drawn attention from both the medical research
community and healthcare policymakers. In recent years, the focus has been on
the development of novel treatments aimed at managing seizures and improving
the quality of life of affected individuals. The rise in clinical trials and
regulatory incentives for orphan drugs has contributed to accelerated drug
development.
The market dynamics are influenced by the growing patient pool, early diagnosis through genetic testing, and enhanced reimbursement frameworks in several regions. Additionally, the involvement of healthcare organizations and advocacy groups has improved patient access to innovative therapies. However, the market continues to face challenges such as high treatment costs and limited curative options. Despite this, technological progress in molecular biology and neurology is fostering a more optimistic outlook for the development of disease-modifying treatments.
With the global burden of epilepsy disorders increasing and healthcare infrastructures strengthening in emerging economies, the Dravet Syndrome market is positioned for steady growth. Stakeholders, including researchers, clinicians, and policymakers, are focusing on improving long-term outcomes through holistic care approaches and personalized medicine. As a result, the Dravet Syndrome market is expected to experience expanded investment, improved diagnosis rates, and an overall enhanced therapeutic landscape in the forecast period.
Definition:
Dravet Syndrome is a
rare, lifelong form of drug-resistant epilepsy that typically begins in infancy
and is characterized by frequent, prolonged seizures often triggered by fever
or high temperatures. It is classified as a developmental and epileptic
encephalopathy due to its profound impact on neurological development and
cognition. The condition is most commonly associated with mutations in the
SCN1A gene, which plays a critical role in the regulation of sodium channels
essential for the normal functioning of neurons. This mutation disrupts the
electrical signaling in the brain, leading to persistent and severe seizure
activity.
The initial signs of Dravet Syndrome typically appear within the first year of life in a previously healthy infant. Early seizures are often generalized tonic-clonic or hemiclonic in nature and may be provoked by vaccinations or febrile illness. As the child grows, different types of seizures such as myoclonic, absence, and atonic seizures may emerge. Over time, the condition can result in developmental delays, motor dysfunction, behavioral issues, and an increased risk of sudden unexpected death in epilepsy (SUDEP).
Due to its complex presentation and resistance to conventional antiepileptic drugs (AEDs), the syndrome poses a substantial therapeutic challenge. The management of Dravet Syndrome involves a multidisciplinary approach encompassing anti-seizure medications, dietary therapies such as the ketogenic diet, behavioral therapies, and supportive care for associated comorbidities. Although not curable, the goal of treatment is to reduce seizure frequency, mitigate developmental regression, and improve the quality of life. The rare nature of the condition, coupled with its severity, has led to increased focus on developing targeted therapies and gaining deeper insights into its genetic underpinnings.
Scope & Overview:
The Dravet Syndrome market encompasses the development, distribution, and
application of therapeutic solutions and supportive technologies aimed at
diagnosing and managing this rare epileptic condition. The market scope
includes pharmacological treatments such as antiepileptic drugs, orphan drugs,
and biologics, as well as non-pharmacological interventions including dietary
modifications, neuromodulation techniques, and supportive therapies. It also
integrates diagnostics and genetic testing services that assist in early and
accurate identification of the syndrome.
With growing understanding of the genetic causes of Dravet Syndrome, the market is witnessing a transition toward precision medicine and gene-targeted approaches. Pharmaceutical innovation has resulted in the introduction of treatments specifically designed for SCN1A mutation-related disorders, distinguishing the Dravet Syndrome market from broader epilepsy therapeutics. In addition, the rising prevalence of genetic testing and personalized care strategies contributes to early intervention and optimized patient outcomes.
The market encompasses key stakeholders such as healthcare providers, researchers, pharmaceutical companies, regulatory bodies, and patient advocacy organizations. Furthermore, it spans diverse geographies, including developed regions with robust healthcare infrastructure and emerging markets with improving diagnostic capabilities. The market’s evolution is heavily influenced by orphan drug legislation, public funding for rare diseases, and collaborative clinical research initiatives.
Overall, the Dravet Syndrome market represents a dynamic and evolving segment of the neurology therapeutics domain. Ongoing research into disease mechanisms, supportive public health policies, and the unmet medical need for effective and accessible treatments continue to shape its trajectory. This landscape offers significant opportunities for innovation, investment, and global health impact over the coming years.
Size
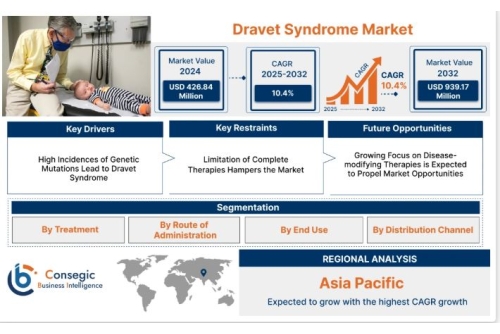
Consegic Business Intelligence analyzes that the Dravet Syndrome market size is growing with a CAGR of 10.4% during the forecast period (2025-2032). The market accounted for USD 426.84 Million in 2024 and USD 463.45 Million in 2025, and the market is projected to be valued at USD 939.17 Million by 2032.
Segmental Analysis:
By Treatment
· Stiripentol
· Cannabidiol
· Fenfluramine
· Clobazam
· Valproate
· Others
Vague Nerve Stimulation OthersBy Route of Administration
Oral InjectableBy End Use
Hospitals Clinics Home care settingsBy Distribution Channel
Offline· Hospital Pharmacies
· Retail Pharmacies
Online Pharmacies
Key Industry Drivers &
Trends:
The Dravet Syndrome market is primarily driven by the increasing prevalence of
genetic and treatment-resistant epilepsy, greater awareness of rare
neurological conditions, and growing support for orphan drug development.
Advancements in genetic diagnostics have facilitated early and accurate
detection of SCN1A mutations, allowing for timely intervention and specialized
treatment plans. This shift toward precision medicine has played a critical
role in shaping current market trends.
Government policies and healthcare initiatives supporting rare disease treatment have also contributed significantly to market growth. Regulatory frameworks in major healthcare markets offer incentives such as fast-track approvals, market exclusivity, and tax credits, encouraging pharmaceutical firms to invest in research and development for Dravet Syndrome therapies. Furthermore, the increasing number of clinical trials focused on novel drug candidates and alternative therapeutic pathways is boosting the pipeline for future treatment options.
One of the prominent trends is the focus on disease-modifying therapies, including gene editing, RNA-based treatments, and novel antiepileptic mechanisms. There is a growing movement away from seizure management alone toward approaches that can alter the course of the disease or address underlying pathologies. Additionally, digital health technologies and telemedicine are becoming integral to managing long-term care, especially in remote or underserved areas.
Collaborative research involving academic institutions, hospitals, and advocacy groups is enhancing the pace of innovation. Meanwhile, patient-centric care models, integrating pharmacological, psychological, and rehabilitative support, are becoming increasingly prevalent. These holistic strategies are redefining how chronic and severe conditions like Dravet Syndrome are approached.
Overall, the convergence of scientific innovation, favorable regulatory landscapes, and multidisciplinary care frameworks continues to drive significant transformation in the Dravet Syndrome market.
Regional Analysis:
The Dravet Syndrome market demonstrates varied growth dynamics across different
global regions, influenced by healthcare infrastructure, regulatory policies,
access to genetic testing, and public awareness. North America represents the
largest market share due to its advanced medical research facilities, strong
presence of pharmaceutical innovation, and supportive regulatory environment.
The U.S., in particular, benefits from robust orphan drug programs, high
investment in rare disease research, and widespread availability of diagnostic
and therapeutic services.
Europe follows closely, with countries such as Germany, France, and the UK spearheading research collaborations and public health programs focused on rare neurological disorders. The European Medicines Agency (EMA) has streamlined regulatory pathways for orphan drugs, and several national healthcare systems offer reimbursements for high-cost rare disease therapies, driving market uptake.
Asia-Pacific is emerging as a promising region, propelled by increasing healthcare spending, improved access to genomic testing, and greater disease awareness. Countries like Japan and Australia have well-established healthcare infrastructures and are investing in rare disease frameworks. Meanwhile, nations such as India and China are gradually building capacity in terms of diagnostics and treatment availability, supported by both public and private initiatives.
Latin America and the Middle East & Africa represent nascent markets, with challenges such as limited diagnostic capabilities, scarce specialist care, and high treatment costs. However, international aid programs and partnerships with global health organizations are beginning to address these gaps.
Overall, regional market expansion depends on a combination of policy support, education initiatives, technological adoption, and economic development. The global push toward equity in rare disease care is expected to enhance access and foster growth in underrepresented regions, creating a more balanced global landscape for Dravet Syndrome management.
Key Players:
Stoke Therapeutics (England)
UCB, Inc. (Belgium)
Lundbeck (Denmark)
Harmonay Biosciences(U.S)
Sanofi(France)
Biocodex, Inc. (France)
Jazz Pharmaceuticals, Inc.(Ireland)
Otter Pharmaceuticals (Subsidiary of Assertio Holdings, Inc.)(U.S)
AbbVie(U.S)
Ovid Therapeutics, Inc.(U.S)
Contact Us:
Consegic Business intelligence
Email : [email protected]
Sales : [email protected]